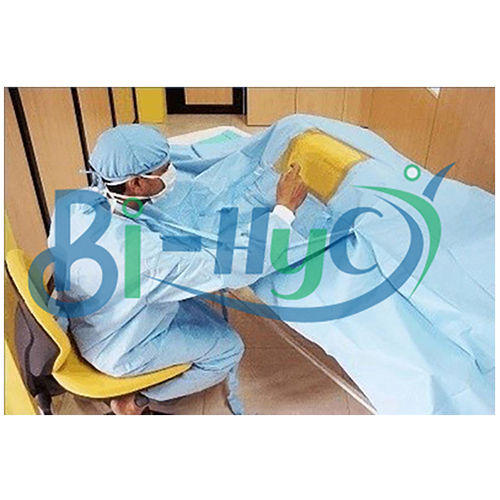
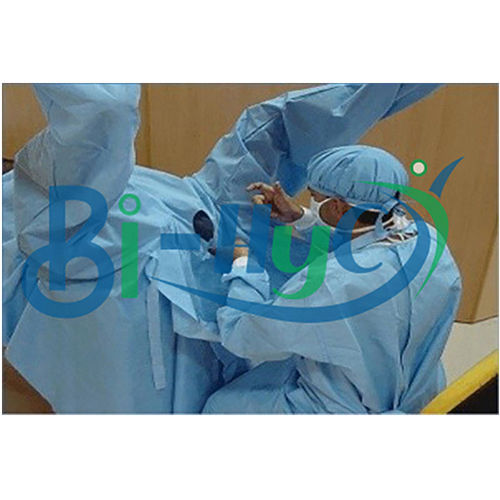
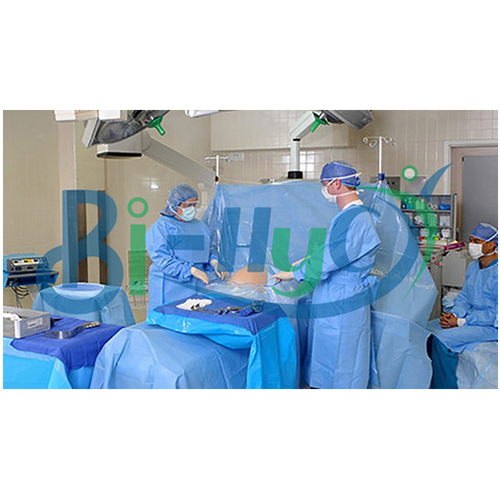
Craniotomy Drape
Price 375 INR/ Piece
Craniotomy Drape Specification
- Sterile
- ETO
- Product Type
- Disposable
- Usage
- Hospital
- Material
- SSMMS
- Size
- 160x300cm
- Color
- Medical Blue
- Application
- Hospital
Craniotomy Drape Trade Information
- Minimum Order Quantity
- 20 Pieces
- Payment Terms
- Paypal, Cash in Advance (CID)
- Supply Ability
- 100 Pieces Per Day
- Delivery Time
- 2 Days
- Sample Available
- Yes
- Sample Policy
- Free samples are available
- Main Export Market(s)
- Asia, Australia, North America, South America, Eastern Europe, Western Europe, Middle East, Central America, Africa
- Main Domestic Market
- All India
- Certifications
- An ISO 9001 & 13485
About Craniotomy Drape
Product details
|
Category |
Standard |
|
Material |
SSMMS |
|
Size |
160x300 cm |
|
Color |
Medical Blue |
|
Gender |
Unisex |
|
GSM |
43 |
|
Packaging Type |
Sterile |
|
Type |
Plain Drapes |
|
Usage/Application |
Neurology Surgery |
Comprehensive Protection During Neurosurgery
Our craniotomy drape is designed to minimize contamination risk during delicate neurosurgical operations. The generous 160x300cm size provides thorough patient and equipment coverage, while the sterile ETO treatment ensures a safe barrier. Suitable for every hospital setting, it delivers peace of mind for surgical teams and patients alike.
Quality Material and Easy Application
Constructed from premium SSMMS fabric, this drape combines strength, softness, and excellent fluid resistance. The medical blue color ensures clarity and identification in surgical environments. Application is straightforward, allowing healthcare professionals to efficiently create a sterile field, thereby enhancing workflow and reducing preparation time.
FAQs of Craniotomy Drape:
Q: How is the Craniotomy Drape applied during neurosurgical procedures?
A: The drape is unfolded and carefully placed over the surgical area after patient preparation. Its 160x300cm size allows for flexible positioning, ensuring the operative site is isolated while maintaining patient comfort and accessibility for the surgical team.Q: What material is used for the Craniotomy Drape and why is it important?
A: This drape is made from SSMMS (Spunbond, Spunbond, Meltblown, Meltblown, Spunbond) fabric, which offers a balance of durability, fluid resistance, and breathability. This ensures a dependable barrier against contaminants during surgery.Q: When should the Craniotomy Drape be used in a hospital setting?
A: It is intended for single-use during neurosurgical or craniotomy procedures to maintain a sterile operating field, ideally used just prior to the commencement of surgery, after the patient and area are properly prepped.Q: Where is the Craniotomy Drape manufactured and supplied from?
A: This product is manufactured in India and distributed both domestically and internationally by our company, serving hospitals, clinics, and healthcare institutions as a trusted supplier, exporter, and trader.Q: What is the sterilization process for this drape?
A: The Craniotomy Drape is sterilized using Ethylene Oxide (ETO), a method that effectively eliminates bacteria, viruses, and fungi without compromising the integrity of the SSMMS fabric. Each batch undergoes stringent quality checks.Q: How does the use of this drape benefit surgical outcomes?
A: Using a high-quality, sterile drape minimizes infection risks by providing an impermeable, clean barrier in the surgical field. This supports better patient recovery and decreases the likelihood of post-operative complications.Q: What distinguishes this drapes usage compared to standard drapes?
A: Its generous size, specific design for neurosurgery, and advanced SSMMS construction make it more effective at fluid management, environmental protection, and surgical site isolation, ensuring optimal conditions during complex procedures.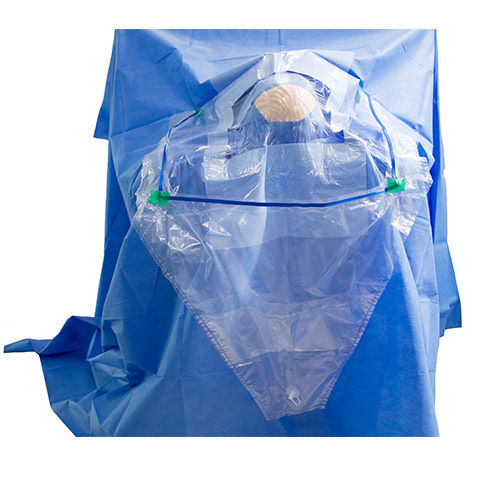

Price:
- 50
- 100
- 200
- 250
- 500
- 1000+
More Products in Medical Disposable Products Category
Hip U Drape
Price 350.00 INR / Piece
Minimum Order Quantity : 20 Pieces
Color : Medical Blue
Application : Hospital
Sterile : ETO
Usage : Hospital
PCNL Drape
Price 280.00 INR / Piece
Minimum Order Quantity : 20 Pieces
Color : Medical Blue
Application : Hospital
Sterile : ETO
Usage : Hospital
TURP Drape
Price 315.00 INR / Piece
Minimum Order Quantity : 20 Pieces
Color : Medical Blue
Application : Hospital
Sterile : ETO
Usage : Hospital
C-Section Drape Kit
Price 430.00 INR / Piece
Minimum Order Quantity : 20 Packs
Color : Medical Blue
Application : Hospital
Sterile : ETO
Usage : Hospital
 |
BHAGWATI LIFE SCIENCE
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese
 Send Inquiry
Send Inquiry